

Curis, Inc. is a therapeutic drug development company focusing on cancer, neurological and dermatological disease indications, with technologies that utilize regulatory pathways that control repair and regeneration. Curis' product development involves the use of small molecules or proteins to modulate these pathways. The company has successfully used this technology and product development approach to produce several promising drug product candidates in the fields of cancer, neurological disorders, hair growth, kidney and other diseases, as well as cardiovascular disease....
+See MoreSharpe-Lintner-Black CAPM alpha (Premium Members Only) Fama-French (1993) 3-factor alpha (Premium Members Only) Fama-French-Carhart 4-factor alpha (Premium Members Only) Fama-French (2015) 5-factor alpha (Premium Members Only) Fama-French-Carhart 6-factor alpha (Premium Members Only) Dynamic conditional 6-factor alpha (Premium Members Only) Last update: Saturday 6 June 2026
2025-10-02 12:31:00 Thursday ET

Stock Synopsis: With a new Python program, we use, adapt, apply, and leverage each of the mainstream Gemini Gen AI models to conduct this comprehensive fund
2018-05-06 07:30:00 Sunday ET

President Trump withdraws America from the Iran nuclear agreement and revives economic sanctions on Iran for better negotiations as western allies Britain,
2019-07-23 09:22:00 Tuesday ET

Harvard economic platform researcher Dipayan Ghosh proposes some alternative solutions to breaking up tech titans such as Facebook, Google, Apple, and Amazo
2019-10-31 13:38:00 Thursday ET
AYA Analytica finbuzz podcast channel on YouTube October 2019 In this podcast, we discuss several topical issues as of October 2019: (1)
2018-06-14 10:35:00 Thursday ET

The Federal Reserve's current interest rate hike may lead to the next economic recession as credit supply growth ebbs and flows through the business cyc
2023-01-09 10:31:00 Monday ET
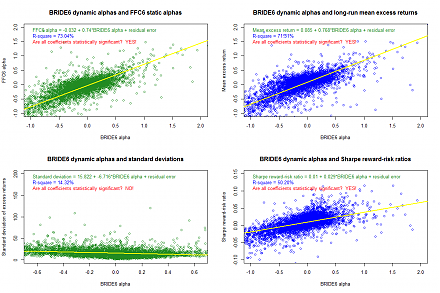
Response to USPTO fintech patent protection As of early-January 2023, the U.S. Patent and Trademark Office (USPTO) has approved our U.S. utility patent